1. Capivasertib in Hormone Receptor-Positive Advanced Breast Cancer
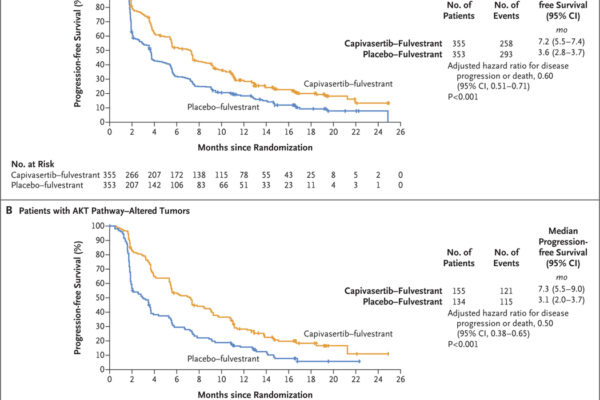
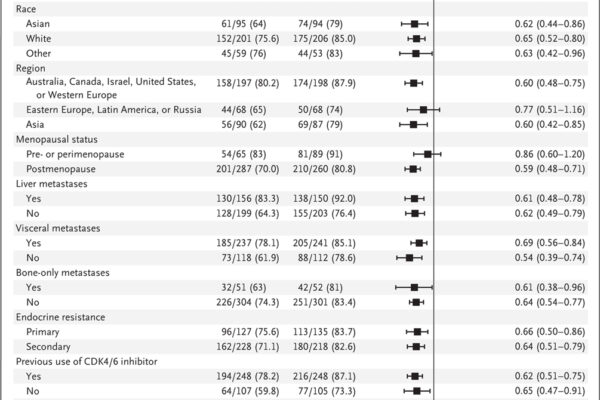
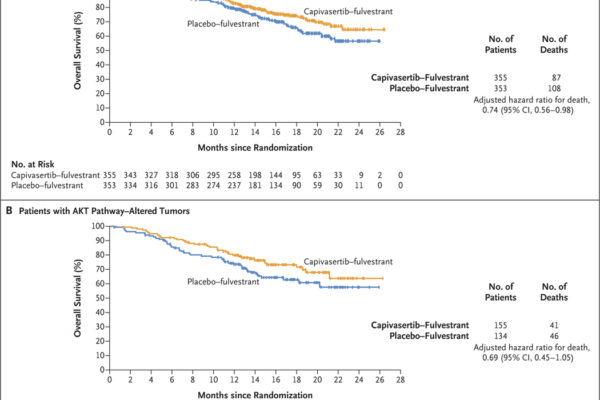
A phase 3 trial assessed capivasertib combined with fulvestrant in patients with hormone receptor-positive, HER2-negative advanced breast cancer. The combination significantly improved progression-free survival compared to placebo plus fulvestrant, with notable benefits in patients harboring AKT pathway alterations.

2. Capivasertib Plus Paclitaxel as First-Line Therapy for Metastatic Triple-Negative Breast Cancer: The PAKT Trial
This randomized phase 2 trial evaluated capivasertib with paclitaxel versus placebo with paclitaxel in untreated metastatic triple-negative breast cancer. The addition of capivasertib led to improved progression-free and overall survival, especially in patients with PI3K/AKT pathway alterations.

3. The Emerging Role of Capivasertib in Breast Cancer
A comprehensive review highlighted capivasertib’s potential across breast cancer subtypes, emphasizing its efficacy in tumors with aberrant PI3K/AKT signaling. Ongoing trials are further exploring its role in combination therapies.

4. Effect of Capivasertib in Patients With an AKT1 E17K-Mutated Tumor: NCI-MATCH Subprotocol EAY131-Y Nonrandomized Trial
This nonrandomized trial focused on capivasertib’s activity in patients with tumors harboring the AKT1 E17K mutation. The study reported a modest overall response rate, suggesting limited single-agent activity in heavily pretreated populations.
Capivasertib: Advancing Breast Cancer Treatment
Capivasertib, an oral AKT inhibitor, has demonstrated significant efficacy in various breast cancer subtypes, particularly those with PI3K/AKT pathway alterations. Clinical trials have shown that combining capivasertib with standard therapies enhances progression-free and overall survival in patients with hormone receptor-positive advanced breast cancer and metastatic triple-negative breast cancer. Ongoing research continues to define its role in targeted cancer therapy, offering hope for more effective and personalized treatment options.
![]()