Precision Biologics and CV
Risk Insights in Autoimmune Diseases
Presented by: Benlysta

The ALTO study, a long-term extension of the APIPPRA phase 2b trial, evaluated the durability of effects from 1-year treatment with abatacept in ACPA-positive individuals with arthralgia but no clinical synovitis who are at risk of developing rheumatoid arthritis (RA). In this randomized, double-blind, placebo-controlled follow-up involving 143 participants (median follow-up 55 months from randomization), the primary outcome—time to clinical synovitis in ≥3 joints, RA diagnosis by 2010 ACR-EULAR criteria, or first DMARD initiation—showed that the initial delay in arthritis-free survival observed during and shortly after treatment persisted significantly up to 4 years. The restricted mean arthritis-free survival time difference was 4.9 months (95% CI 0.1–9.6; p=0.044) at 4 years, though the magnitude diminished over time as survival curves converged thereafter. No significant between-group differences were observed in disease activity measures, patient-reported outcomes, or radiographic progression beyond the initial treatment period.
Participants with a broad autoantibody profile at baseline (e.g., high IgG ACPA titers or positivity across multiple serotypes including rheumatoid factor, IgA ACPA, anti-carbamylated, and anti-acetylated proteins) faced the highest risk of progression but demonstrated the most pronounced and sustained response to abatacept. Safety remained favorable, with 18 serious adverse events in the abatacept group versus 13 in placebo, none deemed related to the study drug. These findings indicate that a limited course of abatacept can delay RA onset for up to 4 years in this high-risk population, particularly among those with more mature autoimmunity, supporting further exploration of targeted interception strategies while highlighting that true prevention requires additional approaches.
The TULIP-SC phase 3 trial, a multinational, randomized, double-blind, placebo-controlled study, evaluated the efficacy and safety of weekly subcutaneous anifrolumab (120 mg) added to standard therapy in 367 adults with moderate to severe systemic lupus erythematosus (SLE) despite ongoing treatment. In the full analysis (anifrolumab n=184; placebo n=183), significantly more patients receiving anifrolumab achieved a BILAG-based Composite Lupus Assessment (BICLA) response at week 52 while maintaining low or reduced oral glucocorticoid doses through week 52 (56.2% vs 34.0%; difference 22.3%, 95% CI 12.3–32.2; P<0.0001), with a faster time to first sustained BICLA response (hazard ratio 2.2, 95% CI 1.5–3.2; P<0.0001). Additional benefits included higher rates of DORIS remission (treatment difference 14.2%, 95% CI 5.6–22.8; P=0.0012) and Low Lupus Disease Activity State attainment (treatment difference 14.1%, 95% CI 4.6–23.6; P=0.0038) at week 52, confirming clinically meaningful improvements consistent with the established intravenous formulation.
Safety profiles were comparable between groups, with serious adverse events occurring in 11.9% of anifrolumab-treated patients versus 10.4% on placebo; herpes zoster incidence was higher with anifrolumab (3.8% vs 1.1%), but overall tolerability remained acceptable with no new safety signals identified. These results support subcutaneous anifrolumab as an effective, convenient self-administration option for patients with active SLE, offering significant disease activity reduction and glucocorticoid-sparing effects over 52 weeks.
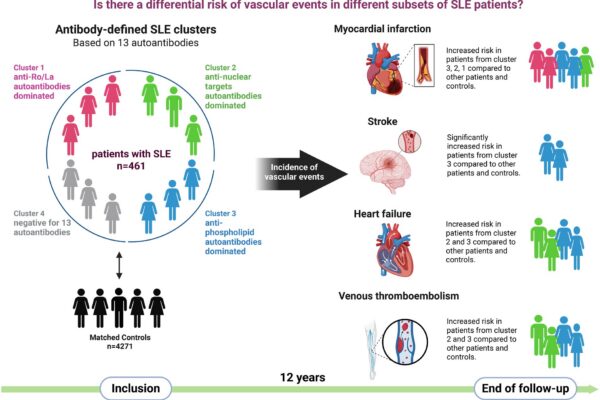
The 12-year prospective cohort study stratified 461 patients with systemic lupus erythematosus (SLE) into four autoantibody-defined clusters and compared their long-term risk of major adverse cardiovascular events (MACE) and venous thromboembolism (VTE) to matched population controls. Cluster 3, dominated by antiphospholipid antibodies (aPL), exhibited the highest relative risk for both MACE (HR 1.91, 95% CI 1.01–3.58) and VTE (HR 2.69, 95% CI 1.05–6.90) compared with reference clusters. Cluster 2, characterized by anti-nucleosome/Sm/RNP/dsDNA antibodies, showed similarly elevated risks for heart failure and VTE despite a younger mean age at baseline. In contrast, cluster 4—negative for all 13 evaluated autoantibodies—demonstrated the lowest incidence of vascular events across the follow-up period (mean 12.2 ? 5.6 years).
These findings highlight distinct vascular risk profiles in SLE based on autoantibody patterns, with the aPL-positive cluster carrying the greatest thrombotic and cardiovascular burden, while autoantibody-negative patients appear relatively protected. The results underscore the clinical utility of autoantibody-based clustering for risk stratification and support intensified surveillance and targeted preventive strategies in high-risk SLE subgroups, particularly those with aPL predominance.
The large-scale analysis of US National Inpatient Sample data from 2006 to 2019 examined trends in acute coronary syndrome (ACS) hospitalizations among patients with systemic lupus erythematosus (SLE) and compared outcomes with non-SLE patients. Among 17,318,554 total ACS hospitalizations, 70,882 (0.41%) involved patients with SLE, who were disproportionately younger (<50 years), female, Black, and had higher prevalences of antiphospholipid syndrome, chronic/end-stage kidney disease, and prior thromboembolism. ACS hospitalization rates declined substantially in both groups—by 40% in SLE patients (primarily from 2015 onward) and by 50% in those without SLE—indicating meaningful progress in cardiovascular risk reduction over the study period.
In-hospital mortality rates were comparable between SLE and non-SLE patients (7.0% vs 6.9%, P=0.52), yet SLE patients experienced significantly longer mean hospital stays (6.22 vs 5.51 days, P<0.001) and higher total charges (US $79,909 vs $74,294, P<0.001), reflecting greater healthcare utilization and resource burden. These findings highlight persistent gaps in ACS outcomes for SLE despite declining hospitalization rates and emphasize the ongoing need for aggressive, targeted cardiovascular risk management in this high-risk population to further reduce morbidity and healthcare costs.
The multicenter observational study of 196 patients with giant cell arteritis (GCA)-associated aortitis compared the real-world effectiveness of tocilizumab (TCZ) administered subcutaneously (SC; n=86) versus intravenously (IV; n=110). Baseline characteristics, including age (mean 69.8 ? 9.4 years), sex distribution (75% women), and inflammatory markers, were comparable between groups. At 24-month follow-up, SC TCZ achieved a slightly higher rate of EULAR-defined remission (83.3% vs 80.6%; P<0.05), while both routes demonstrated equivalent glucocorticoid-sparing effects, rates of imaging remission (assessed by 18F-FDG PET/CT), and absence of systemic inflammation. No significant differences emerged in clinical or imaging outcomes beyond the modest advantage in EULAR remission for the SC formulation.
These findings indicate that subcutaneous tocilizumab is at least as effective as intravenous administration—and potentially marginally superior in achieving formal EULAR remission—in patients with GCA-related aortitis under routine clinical practice conditions. The comparable performance across key secondary endpoints, including imaging improvement and glucocorticoid reduction, supports SC TCZ as a convenient, effective option for managing this severe vascular manifestation of GCA, where aortic involvement carries risks of aneurysm or dissection.